Signature and SDV Overview
Aim
This article briefly describes how to use the “Signature and SDV” overview.
SDV (Source data verification) is only applicable for sponsor users and requires the additional permission “Data Monitor”. All sponsor users with this permission can use the SDV function. Please refer to the article “Monitoring Questionnaires”.
To review the “Signature and SDV” overview a separate permission “Signature/SDV Overview” is necessary.
Procedure
Step 1: Go to the menu section “Study Overview” / subsection “Signature and SDV” (Figure 1).
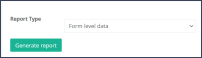
Step 2: Choose a type of report in the dropdown menu between “Form level data” and “Field level data” and click on the green button “Generate report” (Figure 2). • “Form level data”: shows the signatures and SDV information of all questionnaires per patient • “Field level data”: shows the signatures and SDV information of all questions per questionnaire and patient
The Signature and SDV overview can be used to get an overview of the status of signed and/or verificated completed questionnaires and the number of missing signatures. Two actions help to identify further actions:
Sorting a column: By using the two arrow symbol in the right corner of each header, the column can be sorted by its entries, e.g., by name of the user or the number of signatures
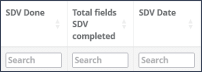
Filtering a column: By typing into the “Search” field below each header, the column can be sorted by its entries, e.g., filter for missing signatures of a transcriber or no SDV done (Figure 3).
The complete Signature and SDV overview table can be downloaded as an Excel file by clicking on “Export” and subsequently “Excel” in the upper right corner.
Updated on 13 Jun 2023
